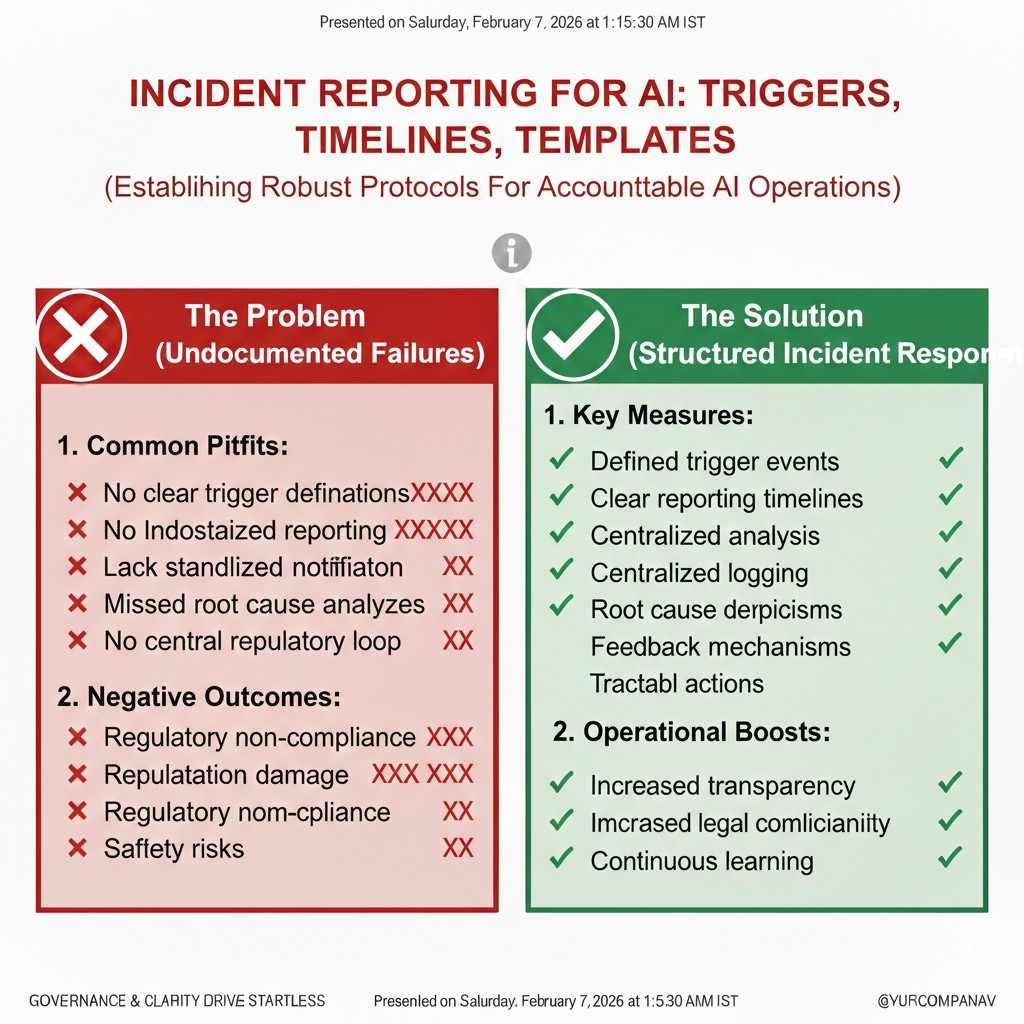
NIST AI RMF for Startups: A Simple Playbook
Most startups build AI fast. That is normal. You ship, you learn, you fix, you ship again. But there is a quiet problem hiding under that speed: risk. Not “big company” risk. Startup risk. The kind that shows up as a surprise customer security review, a blocked enterprise deal, a model that fails in one […]
NIST AI RMF for Startups: A Simple Playbook Read More »